A packaging error is to blame for the mixup that has caused unsuspecting women to consume placebos, when they thought they were taking the birth control pill.
Last month, Baltimore-based drug company, Lupin Pharmaceuticals, recalled a batch of Mibelas 24 Fe tablets after a "confirmed market complaint."
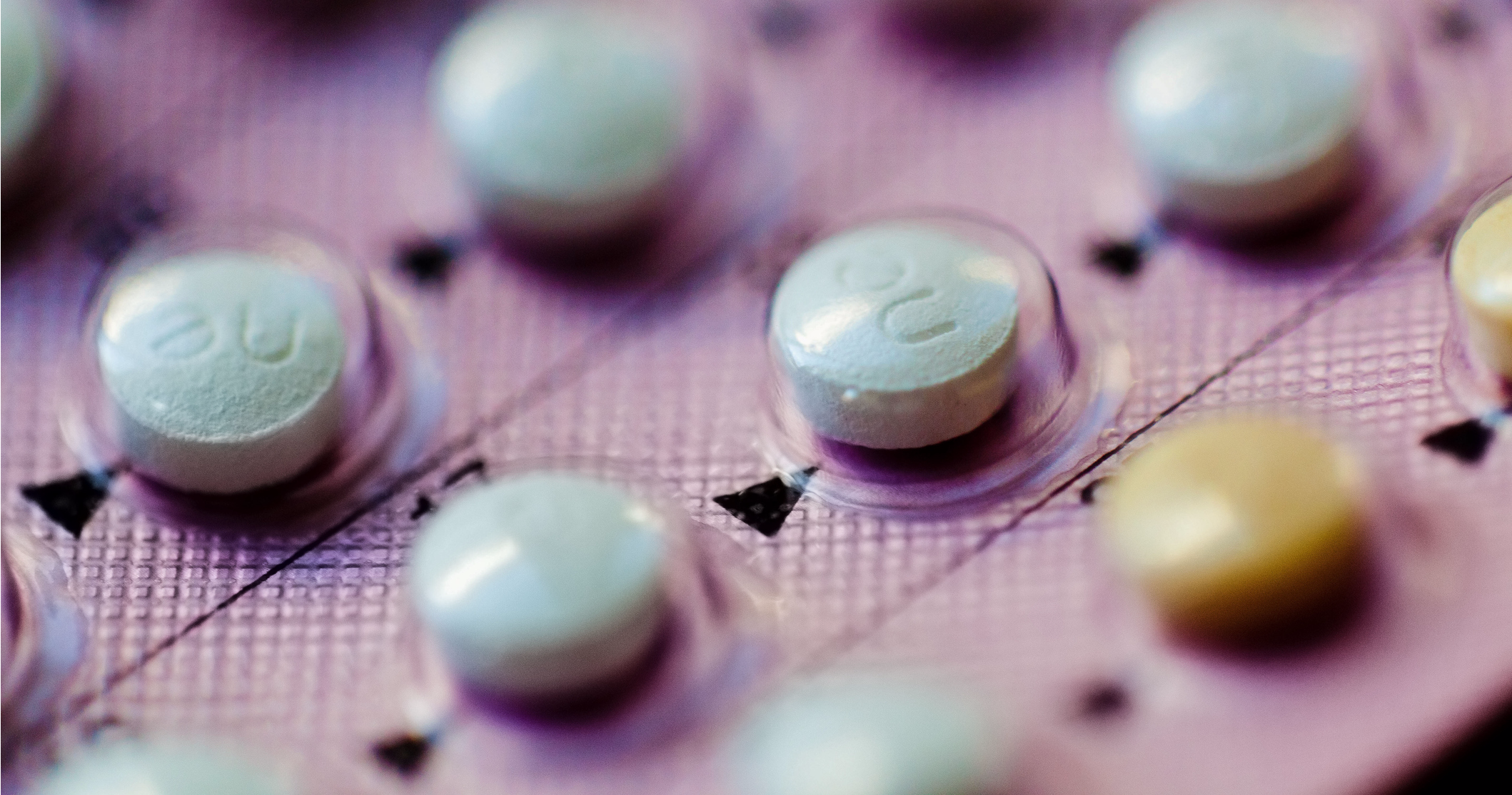
A packaging mix-up switched the contraceptive tablets with the non-hormonal placebos. The package was rotated 180 degrees the wrong way, and inserted into the wallet.
Normally, a box comes with 28 tablets: 24 tablets have active ingredients and are labeled "LU" on one side and "N81" on the other. The four placebos are labeled "LU" on one side and "M22" on the opposite side.
Usually, women would take the placebos at the end of their cycle, when they were on their period. The placebos are a way for patients to keep with the habit of taking their pills daily.
The company released a statement once the recall had been announced:
"As a result of this packaging error, oral contraceptive tablets that are taken out of sequence may place the user at risk for contraceptive failure and unintended pregnancy," the company said. "The reversing the order may not be apparent to either new users or previous users of the product, increasing the likelihood of taking the tablets out of order."
This mistake affects Lot Number L600581 with a May 31, 2018 expiration date.
Although Lupin says that there have been no reported cases of adverse health consequences, anyone affected by this faulty batch is encouraged to alert their doctors about the recalled tablets and return the medication to where you purchased it.
Read more about the recall on the official website of the Food and Drug Administration's website here.
Questions can be directed to Lupin Pharmaceuticals by phone at 1-800-399-2561 between 8:00 a.m. and 5:00 p.m. EST, Monday through Friday.
[h/t Refinery29 / Huffington Post]