If you thought the ongoing recalls of blood pressure medications would end with 2018, we have bad news for you.
Since July, dozens of different generic blood pressure tablets have been recalled because of impurities, and the year is ending on a bad note as one more manufacturer launches their own voluntary recall.

Here's what links all of the recalls from this year together:
- All of the affected tablets feature the ingredient valsartan, or the ingredients losartan and irbesartan.
- All of the recalled medicines are at risk of being tainted with the chemical byproducts NDEA and NDMA, which are thought to increase a patient's cancer risk.
- Most of the affect tablets were made by one of two producers: China's Zhejiang Huahai Pharmaceuticals, or another producer in India that uses a similar manufacturing process.
In our last update about this trend (from earlier this month), we told you that health experts believed the long string of recalls was finally ending.
But pills on the market as recently as this month are being recalled again, so it's important to keep checking your medicine cabinet.
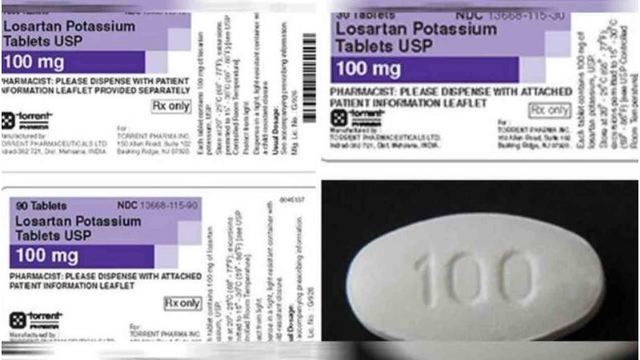
The latest recall comes from Torrent Pharmaceuticals, who are voluntarily pulling two lots of their Losartan potassium tablets (100mg) from shelves.
In an announcement, the FDA said the tablets contain trace amounts of NDEA, which they call a "probable human carcinogen."
As with other recalls of blood pressure medication from this year, the FDA warns patients should continue to take the recalled medication, but contact their doctor or pharmacist to arrange for a replacement.
Suddenly stopping to take the blood pressure medication is considered more dangerous that taking it, even if it your lot is tainted.
If you're not sure what blood pressure medication you're taking, contact your pharmacist.

You can check complete lists of recalled medications containing losartan, valsartan, and irbesartan by clicking the appropriate links on this line.
Make sure to look up your medication to be sure it's safe!
[H/T: CBS News]

