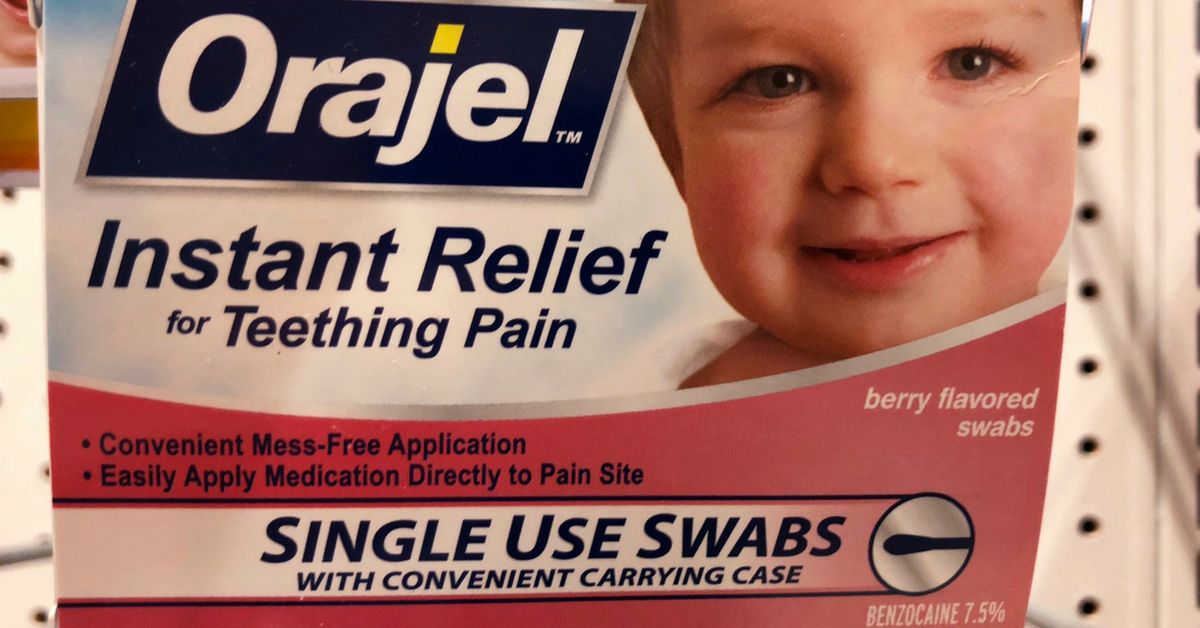
Federal health officials warned parents Wednesday about the dangers of teething remedies that contain a popular numbing ingredient and asked manufacturers to stop selling their products intended for babies and toddlers.
The Food and Drug Administration said that various gels and creams containing the drug benzocaine can cause rare but deadly side effects in children, especially those 2 years and younger.
The agency has been warning about the products for a decade but said reports of illnesses and deaths have continued. Now, it wants teething products off the market, noting there is little evidence they actually work.

"We urge parents, caregivers and retailers who sell them to heed our warnings and not use over-the-counter products containing benzocaine for teething pain," said FDA Commissioner Scott Gottlieb, in a statement.
One major manufacturer, Church and Dwight Co. Inc., said Wednesday it would discontinue its four Orajel teething brands, including Baby Orajel and Orajel Medicated Teething Swabs.
The FDA said it will take legal action against other companies that don't voluntarily comply as soon as possible.
Benzocaine is also used in popular over-the-counter products for toothaches and cold sores in adults, including Orajel and Anbesol and generic drugstore brands.
Products for adults can remain on the market but the FDA wants companies to add new warnings. Church and Dwight will continue to sell its other Orajel products, the company said in a statement.

Benzocaine can cause a rare blood condition linked to potentially deadly breathing problems. The pain-relieving ingredient can interfere with an oxygen-carrying protein in the blood. Symptoms include shortness of breath, headache and rapid heart rate.
The American Academy of Pediatrics does not recommend teething creams because they usually wash out of the baby's mouth within minutes.
Instead, the group recommends giving babies teething rings or simply massaging their gums to relieve pain.
The FDA issued warnings about the teething products in 2006, 2011 and 2014, but it did not call for their removal from the market. Officials reviewed 119 cases of the blood disorder linked to benzocaine between 2009 and 2017, including four deaths, according to the FDA.

Wednesday's action comes more than four years after the consumer advocacy group Public Citizen petitioned the FDA to stop sales of teething products. The agency faced a deadline next week after Public Citizen sued the FDA to force a response to the petition.
Earlier this year, Danielle Kapetanovic from Chantilly, Virginia nearly lost her 15-month-old daughter, Chloe, after she applied a "pea-size" amount of a popular over-the-counter medicine on her gums.
Just seconds after it was applied, Kapetanovic says her daughter went completely limp.
"It was like something had stopped inside of her; like she was not there," she told People. "Even though her eyes were open and locked in a dead stare, she was completely limp. She was turning blue."

Chloe's dad called 911 while Kapetanovic performed CPR. It took about 30 seconds for the infant to wake up again, but her mom described it as "the scariest thing that's ever happened to me."
Since the emergency, the concerned mom has been sharing her daughter's experience so other parents can learn from it.
"Yes, common sense dictates that what happened to my daughter will not happen to every baby, but it could happen to any baby," she wrote on Facebook.
Kapetanovic will without a doubt be among the parents who will be relieved to know that the FDA has taken action against over-the-counter teething gels and creams.
Have you ever used over-the-counter teething medicine on your child? Let us know in the comments!
[H/T: AP]